A federal judge on Tuesday said the Biden administration acted on a “dearth” of information when it approved the practice of mailing the abortion pill, but he gave the Food and Drug Administration more time to clean up the situation.
Judge David Joseph, a Trump appointee, shot down a request by Louisiana to block the mail rules.
He sounded sympathetic to the state’s argument that the FDA was too hasty in 2023 when erased rules requiring in-person dispensing of mifepristone. But he said the agency, now under control of the Trump administration, has promised a rethink and it deserves the space to do that.
“Indeed, given the information available – and, importantly, the dearth of information upon which FDA previously acted to significantly loosen safety restrictions for prescribing mifepristone — the equities and the public interest weigh heavily in favor of FDA completing the job that the law requires it to do,” he wrote in his ruling.
He said he was trusting the FDA to complete a “good faith, evidence-based and expeditious review” of the matter.
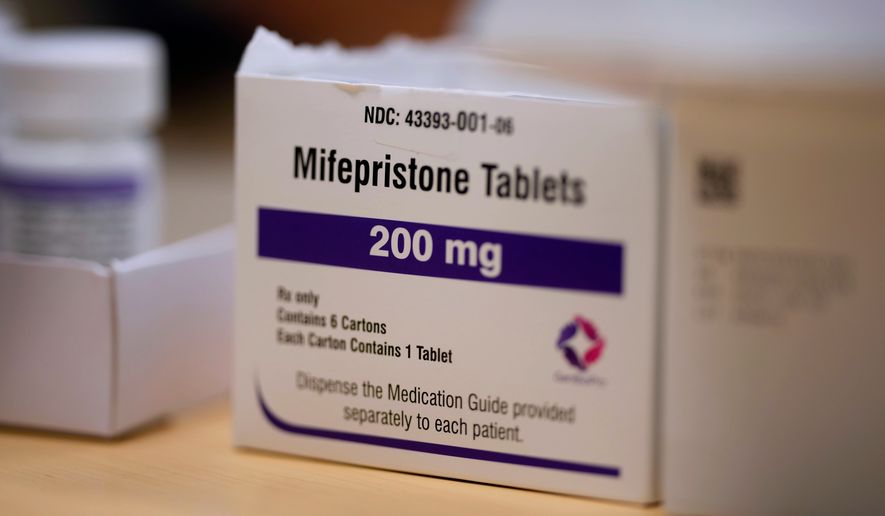
Mifepristole is one of two drugs used in medication abortions, which have become an increasingly common way for women to terminate pregnancies. The ability to mail the pill has been credited for an overall rise in the volume of abortions.
KFF reported 1.06 million abortions in 2023 and 1.14 million in 2024, reversing a trend that had seen the total number decrease.
Pro-life activists have pushed to restrict access to mifepristone, and they have found allies in the Trump administration, which has said it is concerned about reports of “adverse events” in women who used the abortion pill.
The FDA said the approval of mailing the drug was too abrupt.
In Tuesday’s case, Louisiana Attorney General Liz Murrill had challenged the 2023 decision to lift in-person dispensing, clearing the way for the drug to be mailed.
Judge Joseph put the case on hold while the FDA conducts its review.
He asked for an update in six months.
Two firms that manufacture the drug, Danco Laboratories and GenBioPro Inc., had intervened in the case and sought its dismissal altogether. The judge declined to do that.
The American Civil Liberties Union said it was happy the judge didn’t side with Louisiana at this point, but said the FDA’s new review is worrying to those who back abortion rights.
“If the Trump administration limits access to mifepristone, it would fly in the face of science and break President Trump’s campaign promises not to impose new federal restrictions on abortion, which Americans overwhelmingly oppose,” said Julia Kaye, an ACLU lawyer.
Judge Joseph’s ruling came three years to the day after another Trump appointee, Judge Matthew Kacsmaryk, who sits in Texas, issued a ruling suspending the FDA’s approval of mifepristole, which would have taken the drug off the market.
That judge said the approval of the drug for abortion in the Clinton administration was done in violation of FDA rules.
An appeals court stayed much of the ruling, then narrowed it, allowing the initial FDA approval to stand but finding later FDA actions, such as expanding the window for the drug’s use and dropping the in-person requirement, were unlawful.
The Supreme Court in 2024 overturned the case, saying the doctors who had brought the lawsuit lacked standing to challenge FDA decisions.
• Stephen Dinan can be reached at sdinan@washingtontimes.com.



Please read our comment policy before commenting.